![]()
|
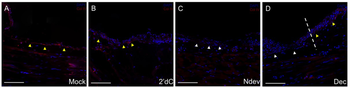
Immunohistochemistry of Anti-Collagen Type IV Antibody Biotin Conjugated. Inhibition of Collagen IV expression in decitabine-treated wound epithelia. Immunohistochemistry staining for Collagen IV, a marker for the basal lamina, in wounds six days post-surgery. Wounds were either untreated (A, mock), received an implanted bead containing 2deoxycytidine without a deviated nerve (B, 2dC), received a surgically deviated nerve to induce formation of an ectopic blastema (C, NDev), or received an implanted bead containing decitabine without a deviated nerve (D, Dec). White arrowheads indicate areas within the wound that are negative for ColIV staining, yellow arrowheads indicate areas that are positive for Col IV staining. Dotted line in (D) indicates the transition between the uninjured skin (right) and the wound (left). Scale bars = 200 microns. |
![]()
|
Immunohistochemistry of Anti-Collagen Type IV Antibody Biotin Conjugated. Microvascular evaluation of the implantation site (IS) with transgenic embryos. Immunostaining for CD34 (cluster of differentiation 34) in ISs of genetically modified trophoblast cells (TC) expressing the control vector (LV-GFP [lentivirusgreen fluorescent protein], A and E, CRISPR-V2, C and G, both 4 dams, 10 ISs), overexpressing tissue TG (TG2) (TG2-LV-GFP, B, 4 dams, 12 ISs), depleted from TG2 (tissue TG, D, 3 dams, 10 ISs), overexpressing FXIII (factor XIII, F, 5 dams, 19 ISs) or depleted from FXIII (H, 4 dams, 12 ISs). CD34 was visualized by cyan fluorescence channel after labeled with Cy5-avidin. Similar exposure time was used for each section and its appropriate control. Quantitative analysis of% CD34 staining in relation to IS area of genetically modified TC overexpressing TG2 (I), depleted from TG2 (J), overexpressing FXIII (K) or depleted from FXIII (L). Image scale bars are 200 µm. Fibrinogen was clearly detected in the anti-mesometrial pole and adjacent to control embryonic TC (Figure 5A, upper part). Increased fibrinogen deposition was detected in IS of FXIII-overexpressing TC, particularly in the IS circumference, while fibrinogen was diminished at the embryonic vicinity (Figure 5B, upper part), as compared with control. CIV localization on the control IS (Figure 5A, lower part) was confined to the anti-mesometrial pole and around the primary decidual zone, while a substantial wider partition was displayed on the FXIII overexpressed TC (Figure 5B, lower part). Inversely to the IS with FXIII overexpressed TC, fibrinogen and Collagen Type IV were hardly detected in IS with FXIII-depleted TC (Figure 5D) relative to control (Figure 5C). |
![]()
|
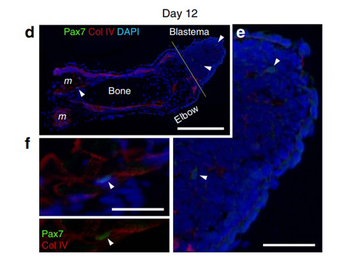
Immunohistochemistry of Anti-Collagen Type IV Antibody Biotin Conjugated. SMFC tracking in larval newt limb regeneration. (d-f) Pax7 immunolabelling of regenerating limbs on day 12 (n 3) after amputation. (d) On day 12, a few Pax7 nuclei (arrowheads) were detected in blastema cells and in satellite cells along the muscle fibers. Col IV, collagen type IV immunoreactivity. DAPI (4, 6-diamidino-2-phenylindole), nuclei. Scale bar, 300 mm. The Pax7 nuclei pointed by arrowheads were enlarged in e and f, respectively. Scale bars, 100 mm. |
![]()
|
Immunohistochemistry of Anti-Collagen Type IV Antibody Biotin Conjugated. SMFC tracking in larval newt limb regeneration. (g) On day 15 when the regenerating part of the limb grew more distally, the number of Pax7 nuclei (arrowheads) in the blastema was dramatically increased. Scale bar, 100 mm. |
![]()
|
Immunohistochemistry of Anti-Collagen Type IV Antibody Biotin Conjugated. SMFC tracking in metamorphosed newt limb regeneration. Merged fluorescence image. Pax7, Col IV, collagen type IV immunoreactivity, To-pro-3: nuclei. (h) Enlargement of a region in the blastema. Scale bars, 250 mm. |
![]()
|
Immunohistochemistry of Anti-Collagen Type IV Antibody Biotin Conjugated. SMFC tracking in metamorphosed newt limb regeneration. Merged fluorescence image. Pax7, Col IV, collagen type IV immunoreactivity, To-pro-3: nuclei. (i) Enlargement of a region proximal to the |