VECTASHIELD Antifade Mounting Medium, 10 ml
Artikelnummer:
VEC-H-1000
- Bilder (4)
| Artikelname: | VECTASHIELD Antifade Mounting Medium, 10 ml |
| Artikelnummer: | VEC-H-1000 |
| Hersteller Artikelnummer: | H-1000 |
| Alternativnummer: | VEC-H-1000 |
| Hersteller: | Vector Laboratories |
| Kategorie: | Zellen/Zellkultur |
| VECTASHIELD Antifade Mounting Medium, 10 ml |
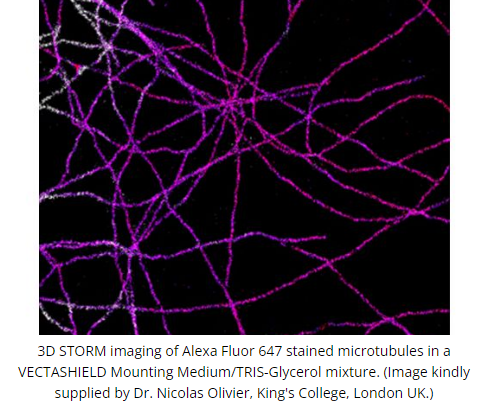
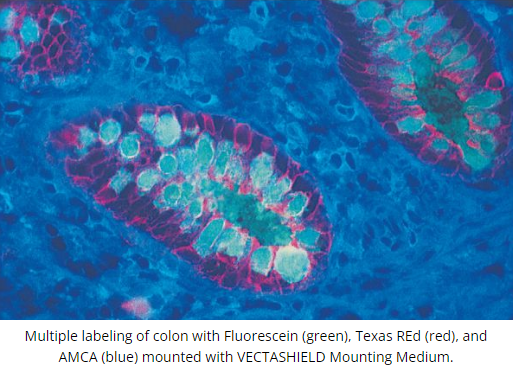
DescriptionVECTASHIELD Antifade Mounting Medium is a unique, stable formula for preserving fluorescence. This medium inhibits rapid photobleaching of fluorescent proteins and fluorescent dyes. Features:
The original VECTASHIELD Mounting Medium does not solidify, but remains a liquid on the slide and can be stored without sealing. If desired, coverslips can be sealed around the perimeter with nail polish or a plastic sealant. Mounted slides should be stored at 4 °C, protected from light.
Specifications
|