![]()
|
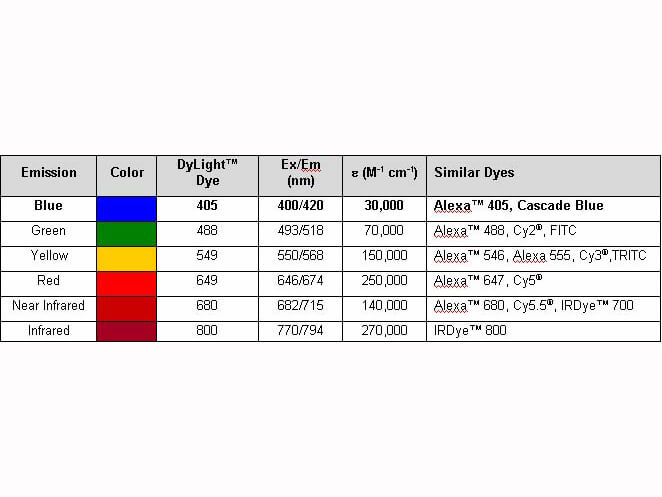
Properties of DyLight(TM) Conjugates. |
![]()
|
Assessment of mitochondrial fusion and fission. B. Representative western blots (original blots are shown in supplementary Fig. S10) and quantification of MFN1/2 and DRP1. No significant changes in the relative levels of proteins that facilitate mitochondrial fusion (MFN1/2) and fission (DRP1) between non-disease (control) and mutant primary fibroblasts. Data are depicted as mean SD, n = 3. The primary antibodies used as follows: MFN1 1:400, MFN2 ( 1:400, DRP1 1:100 and GAPDH 1:30,000 dilutions overnight at 4 C. The membranes were then incubated with fluorescent conjugated secondary antibodies for 1 h, DyLight 800 conjugated goat Anti-Rabbit IgG (611-145-002), Antibody DyLight 680 conjugated Anti-Rabbit IgG made in goat (611-144-003), DyLight 800 conjugated goat Anti-Mouse IgG (610-145-002), and DyLight 680 conjugated donkey Anti-Mouse IgG (610-744-124). Fig 3. PMID: 33725513. |
![]()
|
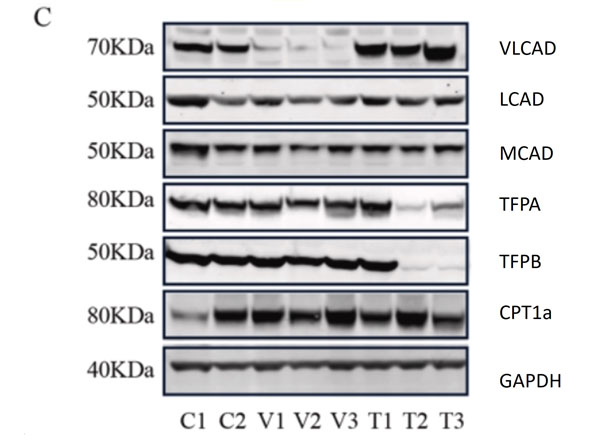
C. Representative western blots, original blots are shown in (supplementary Fig S8-9). And densitometric quantification of relative protein levels from western blots. Data are depicted as mean SD, n = 3, **P < 0.01, ***P < 0.001 and ****P < 0.0001 by one-way ANOVA. Intracellular transport, activation, mitochondrial transport, beta-oxidation, carnitine shuttle, and auxiliary proteins. The primary antibodies used as follows: VLCAD 1:1000, MCAD 1:1000, LCAD 1:1000, TFPa 1:500, TFPb 1:3000, CPT1alpha 1:1000, and GAPDH 1:30,000 dilutions overnight at 4 C. The membranes were then incubated with fluorescent conjugated secondary antibodies for 1 h, DyLight 800 conjugated goat Anti-Rabbit IgG (611-145-002), DyLight 680 conjugated goat Anti-Rabbit IgG (611-144-003), DyLight 800 conjugated goat Anti-Mouse IgG (610-145-002), and DyLight 680 conjugated donkey Anti-Mouse IgG (610-744-124). Fig 1. PMID: 33725513. |
![]()
|
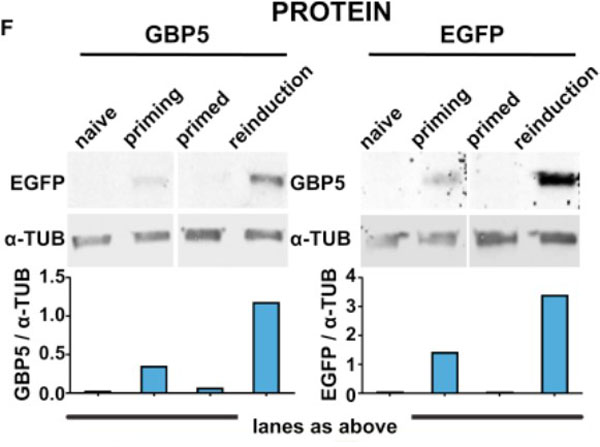
Priming Results in Increased Frequency of Activation and EnhancedGBP5Expression upon Reinduction.(F) EGFP::GBP5 cells were subjected to the IFNgamma treatment regimen outlined inFigure1B, processed for fluorescence western blotting, and probed for GBP5 and EGFP expression. alpha-TUB, loading control. Tubulin-normalized fluorescence intensities are plotted. Fig 3. PMID: 33108759. |
![]()
|
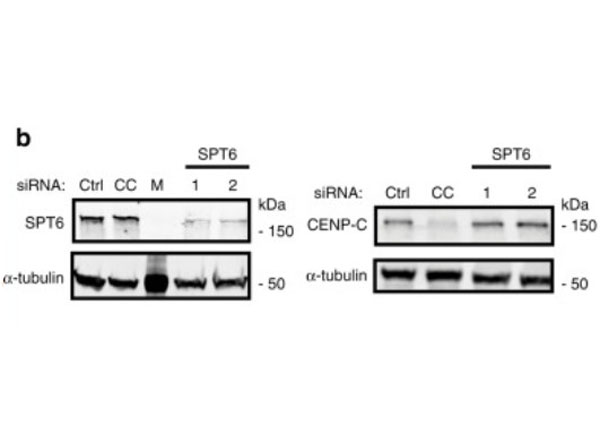
Depletion of human SPT6 leads to the loss of CENP-A maintenance. HeLa cells expressing SNAP-tagged CENP-A were treated with TMR-star to detect previously incorporated CENP-A and siRNA-treated to deplete proteins indicated in (b,c). Cells were then synchronized in S phase by a thymidine block and released. Cells were allowed transit through G1 phase and were collected at the next G1/S boundary by re-addition of thymine.bCells were treated with indicated siRNAs for 48h and extracts were processed for immunoblotting and probed with indicated antibodies. CC CENP-C, M Marker.N=3 independent experiments. Fig 6. PMID: 32522980. |
![]()
|
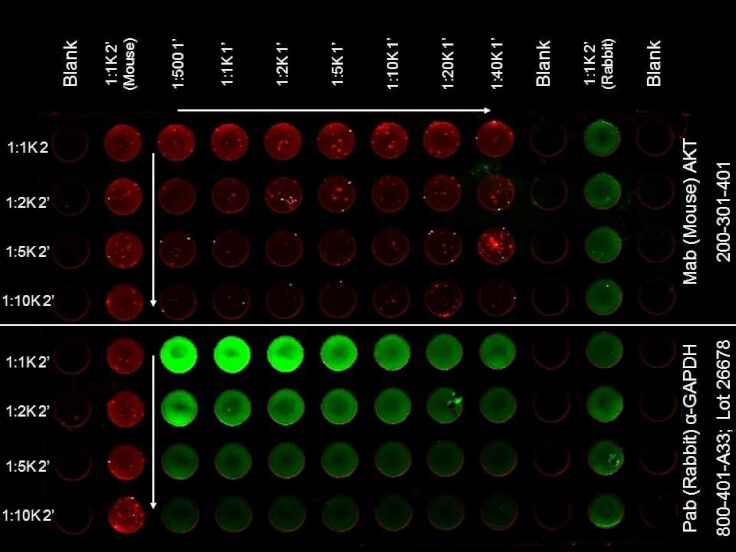
ELISA of DyLight(TM) 680 Conjugated Donkey Anti-Mouse Secondary Antibody. Antigen: HCT-116 cell line. Coating amount: Confluent in the 96 well plate. Primary antibody: AKT or GAPDH antibody at 2 µg/mL. Dilution series: Primary and Secondary Antibodies 2-fold. Mid-point concentration: N/A. Secondary antibody: DyLight(TM) 680 donkey secondary antibody and DyLight(TM) 800 goat secondary antibody starting at 1:1,000. Substrate: None. |
![]()
|
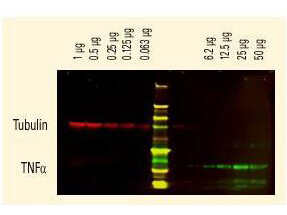
DyLight(TM) dyes can be used for two-color Western Blot detection with low background and high signal. Anti-tubulin was detected using a DyLight(TM) 680 conjugate. Anti-TNFa was detected using a DyLight(TM) 800 conjugate. The image was captured using the Odyssey Infrared Imaging System developed by LI-COR. |
![]()
|
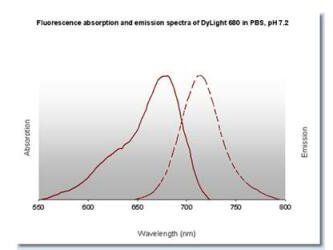
DyLight(TM) 680 Fluorescence Spectra. |