COVID-19 Immunity
Antibody detection by Flow Cytometry
Basis of successfull vaccinations against pathogens as the Coronavirus SARS-CoV-2 is the type, and efficiency of the induced immune response. The human adaptive immune system relies on both, the (antibody-mediated) humoral as well as the T-cell mediated immunity.
Antibodies of diverse isotype and against different SARS-CoV-2 antigens are produced and can be detected and separately identified after a natural COVID-19 infection. Result of a successfull vaccination however is the generation of exclusively antibodies against the spike protein. Existing serology tests detect antibodies to different parts of the virus. To understand if vaccination stimulated an antibody response, a test specifically designed for the antibodies of interest is to be used.
SARS-CoV-2 Multiplex IgG+IgA+IgM kit (CE-IVD)
With their Multiantigen IgG+IgA+IgM Assay (Art. Nr. IMS-IMS0509), our partner Immunostep provides a multiplex, microsphere-based, highly sensitive and specific IVD assay, that measures by flow cytometry the presence or absence of antibodies against four different SARS-CoV-2 antigens simultaneously:
 RBD domain of Spike - detecting SARS-CoV-2 neutralizing antibodies
RBD domain of Spike - detecting SARS-CoV-2 neutralizing antibodies
Spike (S) - identifying immune response after vaccination
Nucleocapsid (N) - identifying immune response to natural infection
Mpro - identifying immune response to viral replication
 Features of the SARS-CoV-2 Multiplex IgG+IgA+IgM Assay:
Features of the SARS-CoV-2 Multiplex IgG+IgA+IgM Assay:
- Exclusive combination of 4 viral antigens analyzed simultaneously
- Wide linearity range
- Near to 100% of sensitivity and specifity
- Reliable and reproducible results
- More results per sample (>12 plex)
- Differentiating natural infection from vaccine response
.jpg)
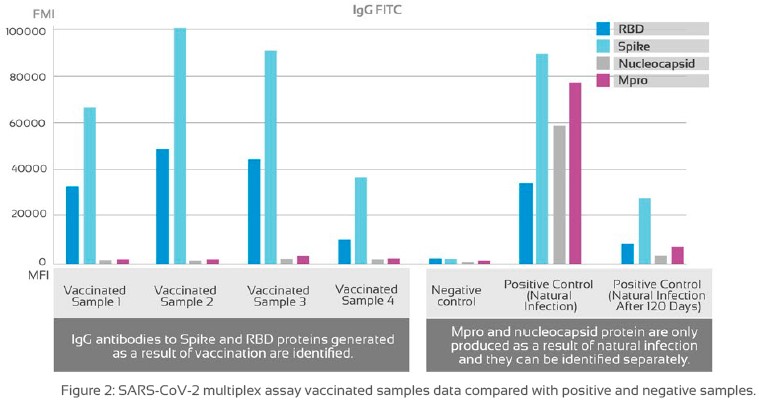
FIG Differentiating natural infection from vaccine response: Pfizer-BioNTech COVID-19 vaccine (Comirnaty) and COVID-19 positive samples were assayed to evaluate IgG antibody response to RBD, S, N and Mpro viral proteins. MFI levels vary from one sample to another, indicating the existance of different antibody response profiles depending on the viral protein analized.
ELISA Kits for SARS-CoV-2 Antibody detection (CE-IVD)
Immunostep provides you with an ELISA option for the detection of different SARS-CoV-2 (COVID-19) antibodies, too:
- SARS-CoV-2 Mpro IgG / IgA ELISA Kit (Art. Nr. IMS-IMS2905 / IMS-IMS2906) - Indirect Enzyme Immunoassay for the detection of IgG Immunoglobulins against Mpro/3CLpro specific SARS-CoV-2 Protein.
- SARS-CoV-2 RBD IgG / IgA ELISA Kit (Art. Nr. IMS-IMS2909 / IMS-IMS2910) - Indirect Enzyme Immunoassay for the detection of IgG Immunoglobulins against RBD SARS-CoV-2 Protein.
- SARS-CoV-2 IgG / IgA Spike ELISA Kit (Art. Nr. IMS-IMS2907 / IMS-IMS2908) - Indirect Enzyme Immunoassay for the detection of IgG Immunoglobulins against Spike SARS-CoV-2 Protein.
Learn more about SARS-CoV-2 (COVID-19) diagnostic ELISA Kits.
21.06.2021

SpinMate Ultrafiltra...
SpinMate Ultrafiltration Concentrators from Protein Ark

Tumor Markers
Recombinant Engineered Antibodies
Antibody-Drug Conjug...
ADC Conjugation Kit from ACROBiosystems

Lipid Nanoparticles
Accelerate your Discovery
Cell Adhesion Molecu...
Key Targets Covered by Biorbyt


