Lipid Nanoparticles
Accelerate your Discovery
Echelon Biosciences a lipid company with decades of hard-won experience. Built on the foundation of extensive expertise in lipids, lipid signaling, and molecular biology, Echelon is not only skilled at chemically synthesizing lipids, but understands how they function in a biological context.
with decades of hard-won experience. Built on the foundation of extensive expertise in lipids, lipid signaling, and molecular biology, Echelon is not only skilled at chemically synthesizing lipids, but understands how they function in a biological context.
Lipid Nanoparticles for RNA delivery
Genetic therapies have been developed for years, with several DNA-based treatments approved. However, these often rely on viral vectors that can trigger immune responses. RNA-based therapies offer a promising alternative and have gained momentum, especially after the success of mRNA vaccines and RNA drugs in recent years.
A key challenge is that naked RNA cannot be directly used, as it is unstable, immunogenic, and poorly taken up by cells. To address this, delivery systems like lipid nanoparticles (LNPs) are used. LNPs protect RNA from degradation and enable its entry into cells, where it can produce therapeutic proteins. Effective therapy depends on careful design of both the RNA and the LNP formulation.
Designing lipid nanoparticles (LNPs) requires balancing effectiveness and immune response. Both mRNA and the ionizable lipids used can trigger immunity—useful for vaccines but problematic for therapies needing repeated dosing, such as gene editing. PEGylated lipids extend circulation time but can induce anti-PEG antibodies, leading to faster clearance. Alternatives like polysarcosine are being explored to reduce this issue without compromising efficiency. A further challenge is targeted delivery: while vaccines benefit from systemic distribution, other therapies need cell-specific targeting. Strategies include modifying LNP surface charge or adding targeting peptides or antibodies to direct them to desired tissues.
RNA Synthesis
Most RNA used in LNPs is produced via in vitro transcription (IVT), a flexible method using different RNA polymerases depending on RNA type and sequence requirements. Key elements such as UTRs, coding regions, ribosome entry sites, and poly(A) tails are included based on the therapeutic goal.
IVT mRNA requires a capping step, and factors like capping efficiency, poly(A) addition, and sequence integrity are evaluated during synthesis and purification. Expression screening is also needed to confirm that the RNA is properly translated and functionally active.
Considerations:
- The type of RNA used in an LNP is the first key design consideration, as siRNA, mRNA, and circular RNA differ in length, structure, and sequence requirements. Even with the minimal necessary elements, additional optimization may be needed depending on the target cell or tissue.
- RNA components are largely determined by RNA type. For mRNA, untranslated regions (5’ and 3’ UTRs) and a poly(A) tail are required to stabilize the transcript and enable translation after delivery. While some elements are broadly applicable, specific UTRs can enhance expression in certain cells, and codon optimization of the coding region can further improve efficiency.
- mRNA cargos require an additional capping step during synthesis. Eukaryotic transcripts naturally carry a 5’ cap, a nucleotide analog that increases stability and is essential for translation. This cap can be added during IVT using synthetic cap analogs or introduced in a later reaction step after transcription. Different cap analogs are available, and their selection involves balancing immunogenicity against translation efficiency.
- RNA synthesis is typically performed using in vitro transcription (IVT), which requires a linear DNA template and specific promoter sequences that enable transcription by recombinant RNA polymerases such as T7. IVT is flexible, allows easy incorporation of modified nucleotides, and is scalable once conditions are optimized. Templates can be generated by restriction digestion, PCR, or oligonucleotide assembly.
- RNA analysis occurs before and after LNP encapsulation. The transcript is first purified by chromatography to remove excess nucleotides and assess purity, followed by verification of correct length and sequence. After confirmation, the RNA is formulated into LNPs, and its integrity is rechecked to ensure no degradation or fragmentation occurred during encapsulation.
LNP formulation
Successful LNP formulation requires several key considerations. The design must account for the target cell or tissue, as well as how effectively ionizable lipids encapsulate and release the cargo. Cargo synthesis should include elements that ensure stability and expression of nucleic acid components during cloning, and may involve modified nucleotides to reduce immunogenicity.
LNP Components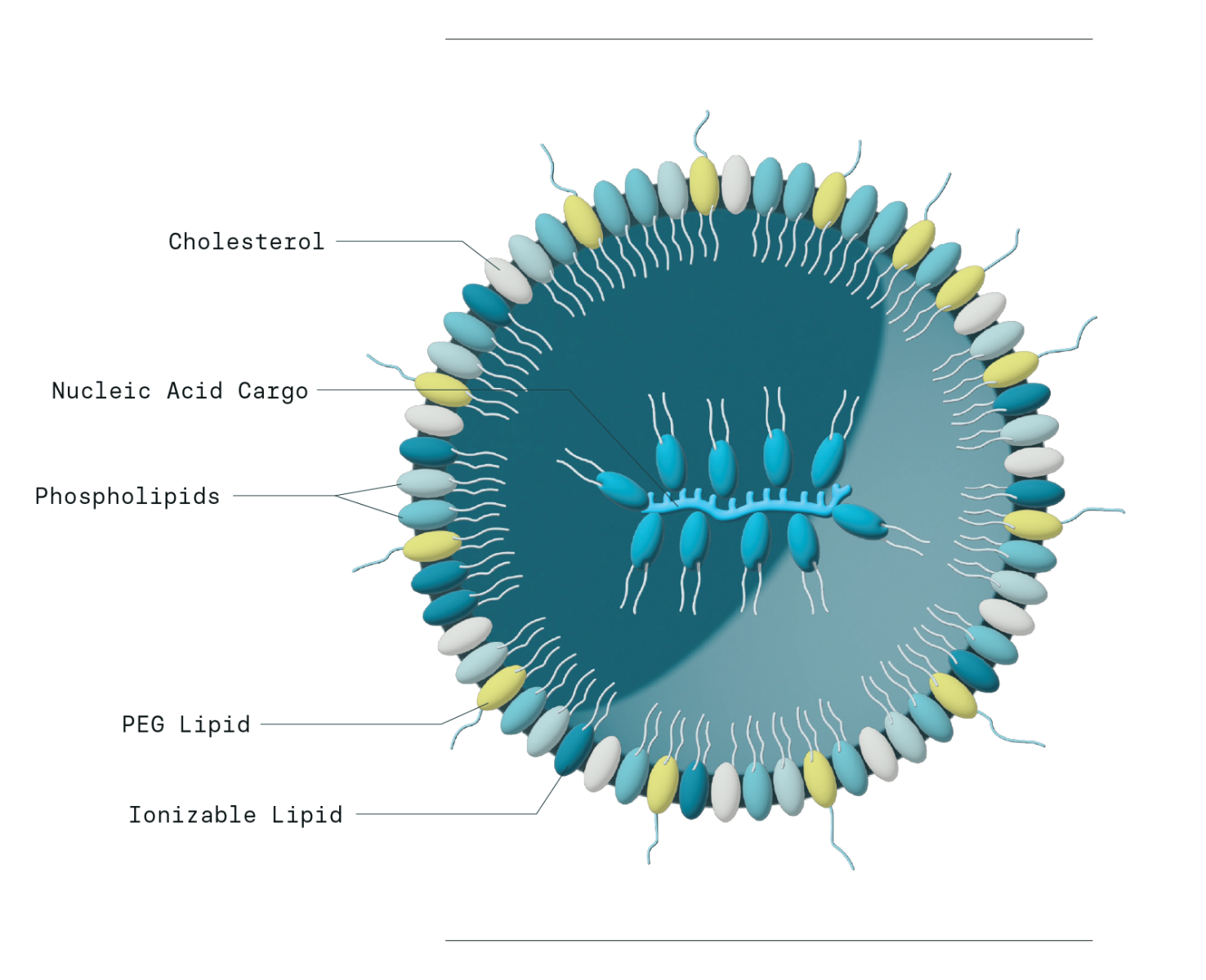
The building blocks of LNPs are remarkably simple. A typical LNP includes an ionizable or cationic lipid, cholesterol, at least one phospholipid, and a polyethylene glycol (PEG) modified lipid. These lipid components are used to shield or encapsulate a nucleic acid cargo that encodes a therapeutic protein.
The type of cargo ranges from mRNA or siRNA to plasmid DNA.
The choice of lipids in the formulation is influenced by the cargo type.
Once sufficient cargo is produced, initial formulation screening evaluates how different ionizable lipids and compositions assemble with the cargo. Downstream analysis provides more than encapsulation data and is essential for assessing formulation success. Finally, expression screening is recommended, since not all formulations that pass analytical checks will produce functional LNPs.
Considerations:
- The key design factor in LNPs is the ionizable lipid, which controls cargo encapsulation and release and must match the type of nucleic acid (e.g., dsDNA, mRNA, siRNA). LNP size and surface charge can be further tuned by adjusting the types and ratios of phospholipids and PEG lipids.
- Cargo synthesis for RNA LNPs is typically done by in vitro transcription from a DNA template. For mRNA, non-coding elements like UTRs are chosen based on the target tissue or cell type to improve stability and translation. Cap structure selection must also be planned in advance, as it may depend on specific sequence requirements at the start of the transcript.
- Once lipids and cargo are prepared, LNP formulation proceeds using automated microfluidic mixers for reproducibility. These systems precisely control rapid mixing of organic-phase lipids with aqueous cargo and allow flexible adjustment of mixing ratios, which can be important for different RNA types.
- Initial LNP analysis focuses on measuring cargo encapsulation. Further characterization includes particle size, molecular weight, surface charge, and polydispersity. Formulation and analysis are iterative processes during screening, and determining whether a formulation is sufficient depends on these key parameters.
- In vitro expression screening is a supplemental step after formulation and analysis. While in vitro and in vivo results may not always align, it provides early validation of LNP performance and can guide further screening or selection for in vivo testing. Using benchmark LNPs with known high expression can improve reliability.
Ionizable and Helper Lipids for LNP Formulation
To support these challenges in LNP development, Echelon Biosciences provides a range of high-quality ionizable and helper lipids for research and development applications.
Ionizable Lipids
Ionizable lipids are the key component to building a fully formed lipid nanoparticle. These specialized lipids obtain a positive charge in acidic pH, allowing them to interact with and encapsulate nucleic acids which contain negative charges via
their phosphate groups. Screening and selection of ionizable lipids is based on the type of nucleic acid cargo, the target tissue and therapeutic application.
The need for new ionizable lipids is growing as the types of cells targeted for therapy and varities of RNA continue to expand. Echelon’s chemists have years of expertise in lipid synthesis and are actively adding new ionizable lipids to our product catalog.
Selected Ionizable Lipids from Echelon Biosciences
| Catalog N° | Ionizabe Lipid |
|---|---|
| ECH-N-1003 | AA3-Dlin |
| ECH-N-1007 | LP-01 |
| ECH-N-1010 | 306Oi10 |
| ECH-N-1017 | DOG-IM4 |
| ECH-N-1025 | 6,6’-trehalose dioleate |
| ECH-N-1050 | YSK05 |
| ECH-N-1110 | Lipid 10 |
| ECH-N-1128 | 306-O12B |
| ECH-N-1220 | C12-200 |
| ECH-N-1316 | 306-N16B |
| ECH-N-1319 | Lipid 319 |
| ECH-N-1438 | 4A3-SC8 |
Helper Lipids
Helper lipids refer to the remaining lipid components of LNPs outside of the ionizable lipid. These typically include a combination of cholesterol, a phospholipid, and a PEGylated lipid to build the outer membrane structure that fully encloses nucleic acids captured by the ionizable lipids. Stability and other physiochemical properties of LNPs can be tuned and refined by adjusting the type and ratio of these lipids across LNP formulations.
Selected Helper Lipids from Echelon Biosciences
| Catalog N° | Helper Lipid |
|---|---|
| ECH-L-0112 | 9(10)-Nitrooleic Acid |
| ECH-L-1122 | PLPC (16:0/18:2 PC) |
| ECH-L-1182 | DOPC (18:1/18:1 PC) |
| ECH-L-1618 | POPC (16:0/18:1 PC) |
| ECH-L-2116 | DPPE (16:0/16:0 PE) |
| ECH-L-2118 | DSPE (18:0/18:0 PE) |
| ECH-L-2182 | DOPE (18:1/18:1 PE) |
| ECH-L-2810 | DSPE-PEG2000-Maleimide |
| ECH-L-2811 | DSPE-PEG2000-NH2 |
| ECH-L-2814 | DSPE-PEG2000-COOH |
| ECH-L-6004 | 7-Dehydrocholesterol (7-DHC) |
| ECH-L-6005 | Ergosterol |
| ECH-L-6012 | Cholesterol |
Trailblazer LNP Kits
Echelon’s Trailblazer LNP Kits are designed to simplify early stage exploration of LNPs. Each kit provides a buffer for diluting nucleic acids and the lipids necessary to form LNPs either lyophilized and pre-mixed.
The pre-mixed LNP kit utilizes a standardized molar percentage for each lipid so that accurate comparisons can be made. The lyophilized LNP kit allows the experimenter to adjust the ratio of the lipid components as needed.
| Catalog N° | Product |
|---|---|
| K-6300L | TRAILBLAZER |
| K-6300M | TRAILBLAZER PRE-MIXED |
If you have a novel ionizable or cationic lipid structure you would like to screen or if an item is not listed in our catalog, please feel free to contact us for advice and custom synthesis
05.05.2026

SpinMate Ultrafiltra...
SpinMate Ultrafiltration Concentrators from Protein Ark

Tumor Markers
Recombinant Engineered Antibodies
Antibody-Drug Conjug...
ADC Conjugation Kit from ACROBiosystems
Cell Adhesion Molecu...
Key Targets Covered by Biorbyt
Single-Domain Antibo...
Superior staining performance with Biotium MiniMab™

